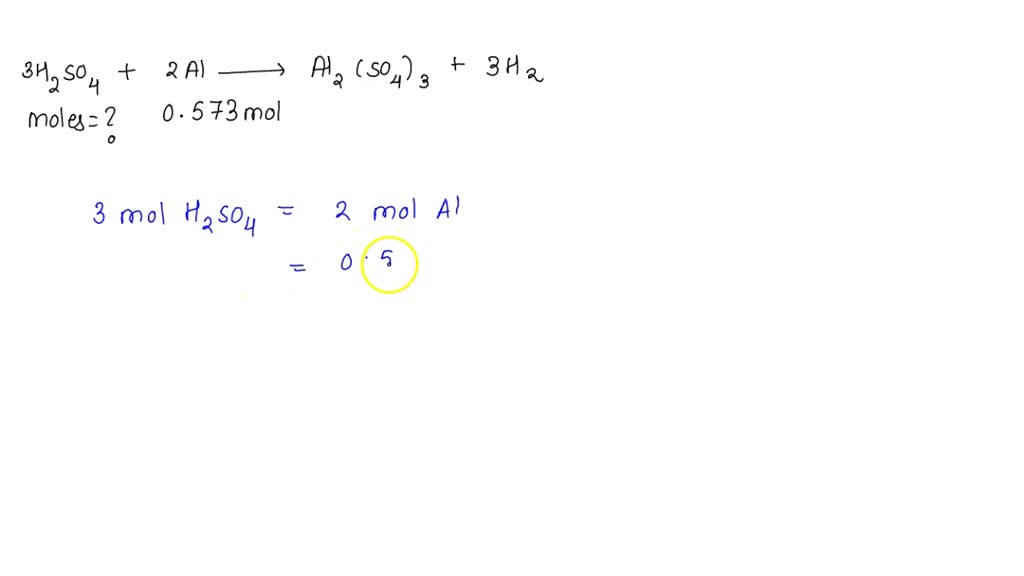
SOLVED: 3 H2SO4(aq) + 2Al(s) = Al2(SO4)3(aq) + 3H2(g) how many moles of sulfuric acid are consumed if 0.573 moles of aluminum reacts according to the reaction shown above? the molar mass

Sulfuric Acid, Sulphuric Acid, Is A Highly Corrosive Strong Mineral Acid With The Molecular Formula H2SO4, Vector 3d Molecular Structure Royalty Free SVG, Cliparts, Vectors, And Stock Illustration. Image 78444709.

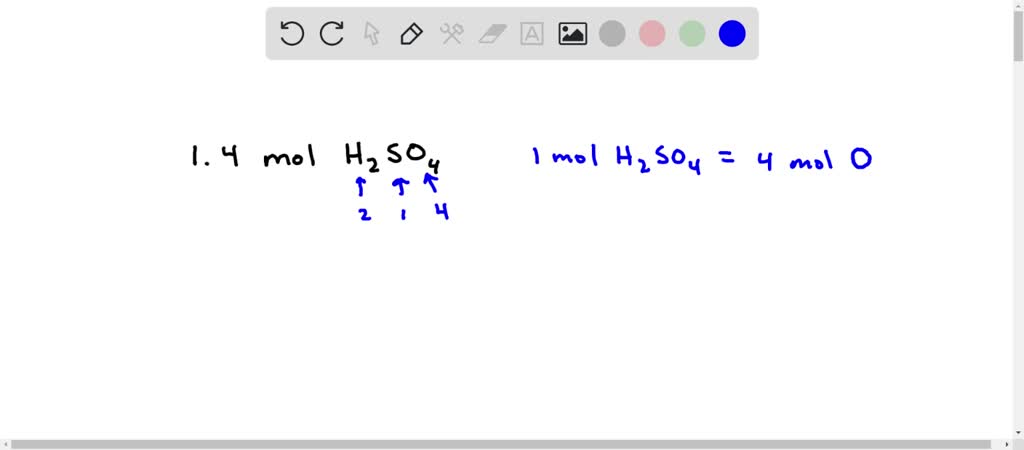
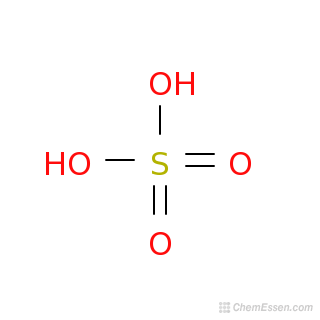
The molecular mass of H2SO4 is 98 amu. Calculate the number of moles of each elements in 294 g of H2SO4
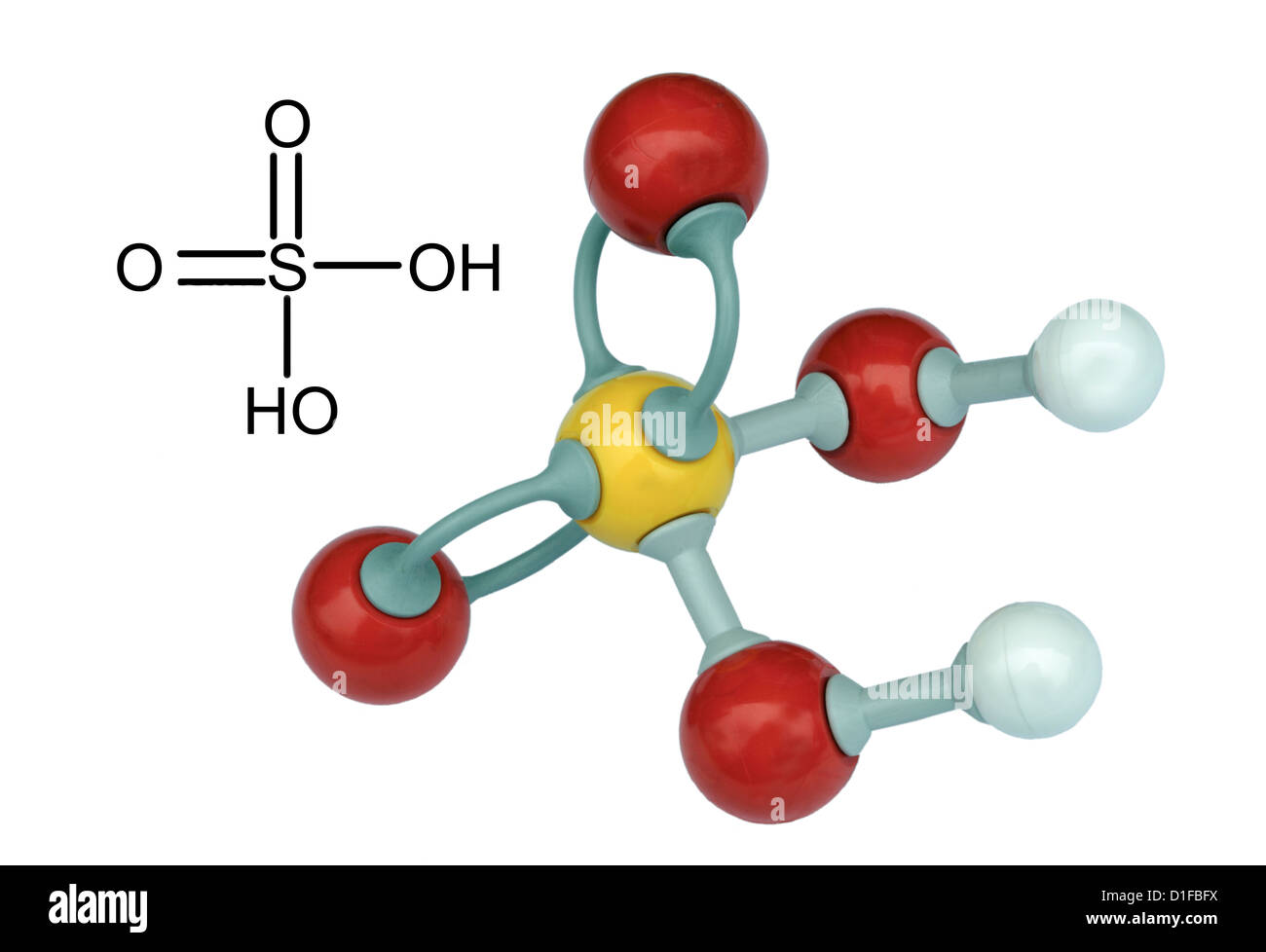
Sulfuric acid, H2SO4, ball-and-stick model, molecular and chemical formula with binding lengths. Known as sulphuric acid, or oil of vitriol Stock Photo - Alamy
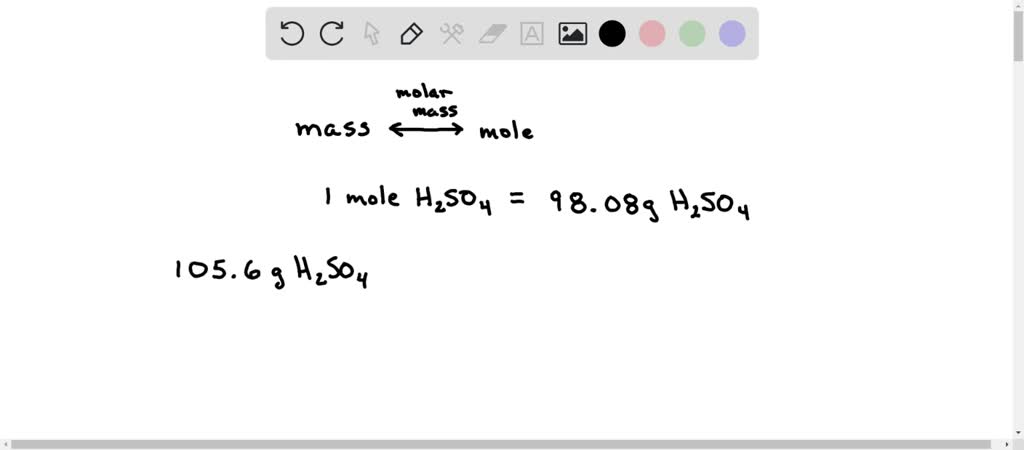





![Sulfuric Acid [H2SO4] Molecular Weight Calculation - Laboratory Notes Sulfuric Acid [H2SO4] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2021/11/sulfuric-acid-molecular-weight-calculation.jpg)