Oxidation of secondary alcohols using 10 mol% lanthanum triflate as... | Download Scientific Diagram

SOLVED: a mole of sodium atoms contains 6.02 x 10^23 atoms. How many moles would be needed in order to have 2.50 x 10^24 atoms?
What are some important formulas for mole concepts, like how to find the number of atoms, etc.? - Quora
Mole Calculations. ? Visualizing a Chemical Reaction Na + Cl 2 NaCl ___ mole Cl 2 ___ mole NaCl___ mole Na ppt download

How many atoms or molecules are present in 1.0 mol of Au? How am I able to find the answer? | Socratic
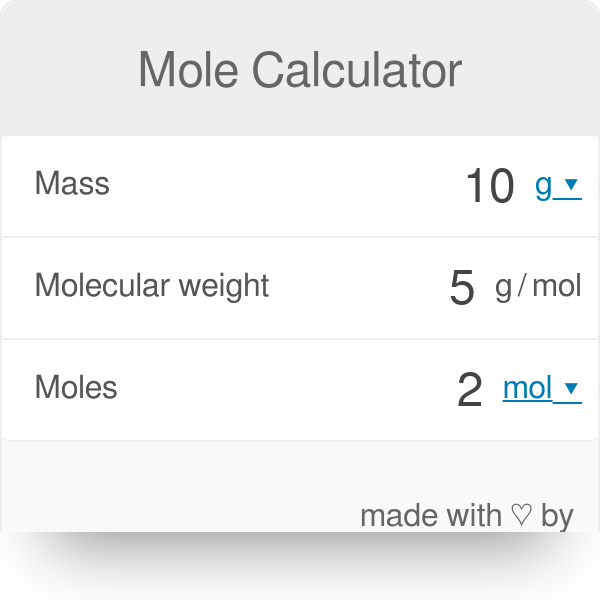
4 A-3 The Mole The mole (abbreviated mol) is the SI unit for the amount of a chemical species. It is always associated with a ch
![For the reaction: 2A + B → A2B , the rate = k[A][B]^2 with k = 2.0 × 10^-6 mol^-2 L^2 s^-1 . Calculate the initial rate of the reaction when [A] = For the reaction: 2A + B → A2B , the rate = k[A][B]^2 with k = 2.0 × 10^-6 mol^-2 L^2 s^-1 . Calculate the initial rate of the reaction when [A] =](https://d1hhj0t1vdqi7c.cloudfront.net/v1/TVhJa2FqVWNSMFk=/sd/)
For the reaction: 2A + B → A2B , the rate = k[A][B]^2 with k = 2.0 × 10^-6 mol^-2 L^2 s^-1 . Calculate the initial rate of the reaction when [A] =

Quantos atomos de oxigenio existem em 0,10 mol de glicose (C6H1206)? Constante de avogadro: 6,x - Brainly.com.br